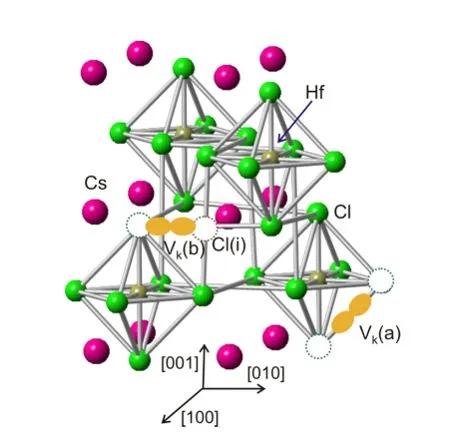
Single crystals of Cs2HfCl6 have recently been identified as promising scintillating material. Furthermore, extended first-principles calculations of Cs2HfCl6 electronic and optical properties have been reported, which, among others, indicate the self-trapping of holes and electrons in the form of VK and polaron centers, respectively. Renewed interest in halide scintillators appeared in the late 1990s when rare-earth-based halides were studied and later on the Ce-doped LaCl3, LaBr3, and CeBr3 were discovered as scintillators with both the high light yield and excellent energy resolution. Further optimization of the LaBr3:Ce by divalent ion codoping provided the material with the best energy resolution within single-crystal scintillators. Moreover, a growing interest in materials from the elpasolite group, such as Cs2LiYCl6:Ce, Cs2LiLaCl6:Ce, Cs2NaLaBr6:Ce, Cs2NaLaI6:Ce, and Cs2LiLaBr6:Ce, and those with mixed anions has arisen.(10, 11) Among them, the Li-containing compositions can also be considered for thermal neutron detection. Most recently, SrI2:Eu single crystal was rediscovered, having ultrahigh light yield and excellent energy resolution as well. Similarly, the LuI3:Ce single crystals have been repeatedly addressed and found to be very competitive in the view of achievable light yield. All of the above-mentioned materials are highly hygroscopic, which makes their practical application difficult and expensive.