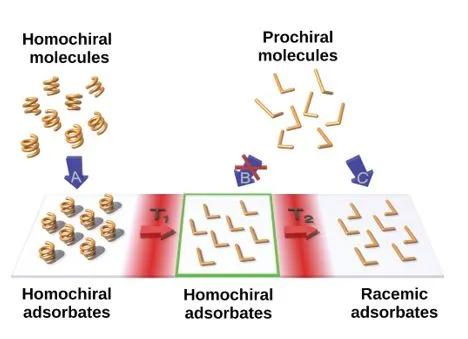
The chirality of molecular structures is paramount in many phenomena, including enantioselective reactions, molecular self-assembly, biological processes and light or electron-spin polarization. Flat prochiral molecules, which are achiral in the gas phase or solution, can exhibit adsorption-induced chirality when deposited on surfaces. The whole array of such molecular adsorbates is naturally racemic as spontaneous global mirror-symmetry breaking is disfavoured. Here we demonstrate a chemical method of obtaining flat prochiral molecules adsorbed on the solid achiral surface in such a way that a specific adsorbate handedness globally dominates. An optically pure helical precursor is flattened in a cascade of on-surface reactions, which enables chirality transfer. The individual reaction products are identified by high-resolution scanning-probe microscopy. The ultimate formation of globally non-racemic assemblies of flat molecules through stereocontrolled on-surface synthesis allows for chirality to be expressed in as yet unexplored types of organic–inorganic chiral interfaces.